- Products / Technology
- Technology
- SDGs /CSR /Environment
- NEWOct. 06, 2005
Development of a "Super Intelligent Catalyst" that Regenerates All of Its Precious Metals
Oct. 06, 2005
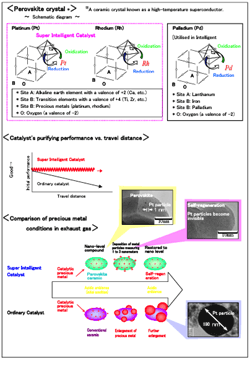
- Dramatically reduces the amount of precious metals used in automotive catalysts - Daihatsu Motor Co., Ltd., working in cooperation with the Japan Atomic Energy Agency (JAEA), Cataler Corporation, and Hokko Chemical Industry Co., Ltd., recently developed a "Super Intelligent Catalyst." The Super Intelligent Catalyst provides all three of the most commonly used precious metals (palladium, platinum and rhodium) with a self-regenerative function, thus successfully minimizing the degradation of the catalyst's exhaust gas purification performance. This technology significantly reduces the consumption of precious metals, which are valuable natural resources, and also achieves clean emissions at an ultra-low cost. The Super Intelligent Catalyst has realised the highest levels of resource conservation and cost reduction possible with present-day technology, and is expected to become the global standard for automotive catalysts. The original "Intelligent Catalyst," which was industrialised in 2002, gave a self-regenerative function to palladium, a precious metal whose exhaust gas purifying activity deteriorates and for which a reduction in consumption was considered most difficult. The technology achieved a major reduction in the amount of palladium used in automotive catalysts, and also lowered the cost of the catalyst. By the end of September 2005, the number of vehicles equipped with the original Intelligent Catalyst had exceeded 1,500,000 units. The Super Intelligent Catalyst has been developed from the basic concept of the Intelligent Catalyst to an even higher level. By combining new materials that are completely different from those used to enable the self-regenerative function in palladium, Daihatsu succeeded in giving a self-regenerative function to platinum and rhodium.
Principle of the self-regenerative function
The Super Intelligent Catalyst has the capability to sense ambient changes, adapt its structure and function, and perform appropriately at all times. The metal ions of platinum and rhodium are replaced in a special perovskite-type ceramic crystal lattice, and surrounded by six ions of oxygen. Platinum and rhodium react in response to an inherent fluctuation in the oxygen concentration of exhaust gas from a gasoline engine. Platinum and rhodium come out of the perovskite-type ceramic crystal and form metallic nano-particles when there is insufficient oxygen. Conversely, when there is surplus oxygen, the metallic nano-particles of platinum and rhodium return to the perovskite-type crystal as metal ions. This motion is repeated according to the natural fluctuation of the exhaust gas, and is called the self-regenerative function. * In a gasoline engine, the air-to-fuel ratio is constantly monitored and adjusted to optimize combustion. As a result, the fluctuation between reduction and oxidation (redox) in the exhaust gas occurs at a cycle of several times per second (1-4 Hz). The motion of platinum and rhodium in and out of perovskite-type crystal occurs continuously, and the particle growth of platinum and rhodium is suppressed by the self-regenerative function. As a result, the Super Intelligent Catalyst is able to maintain excellent catalytic performance using only a small amount of precious metals. The original Intelligent Catalyst with a self-regenerative function for palladium (Pd) was developed in 2002, and it contained lanthanum and iron as the LaFePdO3 perovskite. However, when the LaFeO3 perovskite crystal is used for platinum and rhodium, since the stable valences differ from Pd, the self-regeneration does not function well. The new Super Intelligent Catalyst provides a perfect self-regenerative function for platinum and rhodium thanks to the development of a completely new perovskite-type crystal consisting of a different composition.
References
Nishihata, Y. et al., Self-regeneration of a Pd-perovskite catalyst for automotive emissions control. Nature 418, 164-167 (2002).
<a href="05100601_02.pdf" target="_blank">Magnified View</a>
The Japan Atomic Energy Agency cooperated in atomic level analysis using the synchrotron radiation of SPring-8 to elucidate the self-regenerative function. The Agency played a key role in determining the overall direction of scientific efforts by conducting an atomic-level analysis of the entry and exit (solution and deposition) of precious metals into and out of perovskite oxides. This analysis was carried out using X-ray radiation produced in a large-scale radiation facility called SPring-8.<br><br> <a href="/news/2005/05100601.html" target="_blank">Supplementary Information</a>






